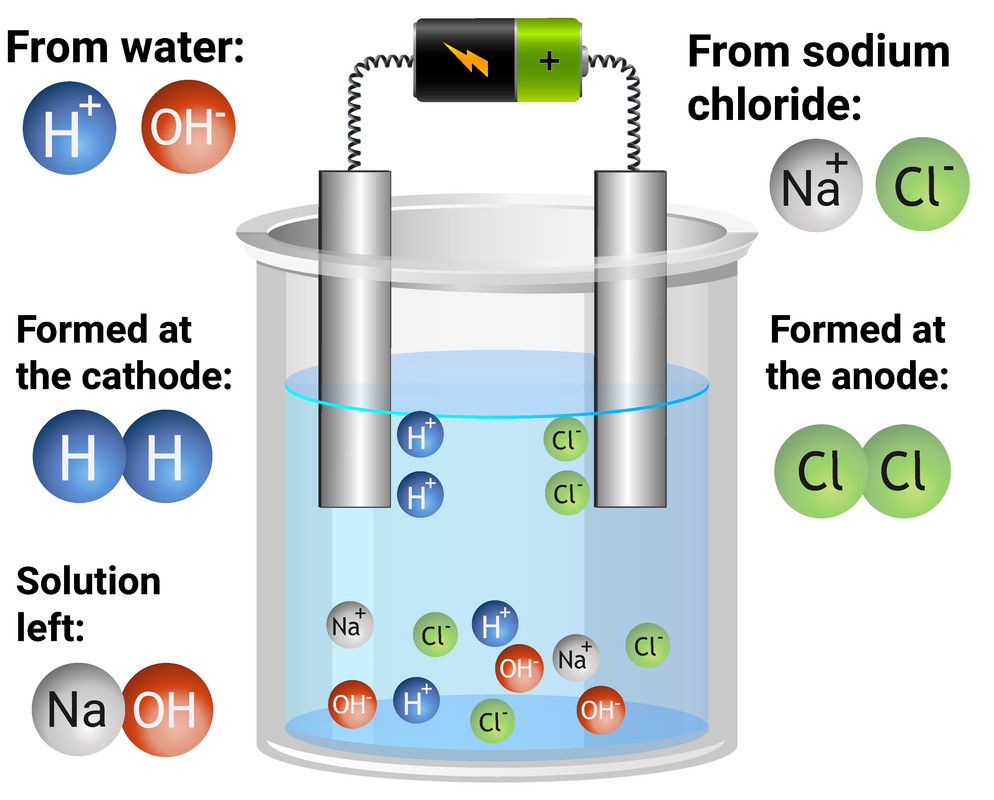
Electrolysis Chem Libre . Describe the process of electrolysis. Describe the process of electrolysis. electrolysis is a process by which electrons are forced through a chemical cell, thus causing a chemical. By the end of this section you will be able to: The opposite is true for electrolytic cells. Compare the operation of electrolytic cells with that of galvanic cells. in electrolysis, an electric current it sent through an electrolyte and into solution in order to stimulate the flow of. in galvanic cells, chemical energy is converted into electrical energy. using electricity to force a nonspontaneous process to occur is electrolysis.
from www.revisechemistry.uk
in galvanic cells, chemical energy is converted into electrical energy. Compare the operation of electrolytic cells with that of galvanic cells. in electrolysis, an electric current it sent through an electrolyte and into solution in order to stimulate the flow of. Describe the process of electrolysis. The opposite is true for electrolytic cells. electrolysis is a process by which electrons are forced through a chemical cell, thus causing a chemical. Describe the process of electrolysis. using electricity to force a nonspontaneous process to occur is electrolysis. By the end of this section you will be able to:
Electrolysis OCR Gateway C3 revisechemistry.uk
Electrolysis Chem Libre using electricity to force a nonspontaneous process to occur is electrolysis. Compare the operation of electrolytic cells with that of galvanic cells. electrolysis is a process by which electrons are forced through a chemical cell, thus causing a chemical. in galvanic cells, chemical energy is converted into electrical energy. Describe the process of electrolysis. Describe the process of electrolysis. By the end of this section you will be able to: The opposite is true for electrolytic cells. using electricity to force a nonspontaneous process to occur is electrolysis. in electrolysis, an electric current it sent through an electrolyte and into solution in order to stimulate the flow of.
From www.aakash.ac.in
Electrolysis Definition, Mechanism, Meaning & Application Chemistry Electrolysis Chem Libre in galvanic cells, chemical energy is converted into electrical energy. using electricity to force a nonspontaneous process to occur is electrolysis. Compare the operation of electrolytic cells with that of galvanic cells. Describe the process of electrolysis. electrolysis is a process by which electrons are forced through a chemical cell, thus causing a chemical. By the end. Electrolysis Chem Libre.
From www.slideserve.com
PPT Electrolysis An Introduction VCE Chemistry Unit 4 Chemistry at Electrolysis Chem Libre Describe the process of electrolysis. in galvanic cells, chemical energy is converted into electrical energy. electrolysis is a process by which electrons are forced through a chemical cell, thus causing a chemical. Describe the process of electrolysis. Compare the operation of electrolytic cells with that of galvanic cells. The opposite is true for electrolytic cells. in electrolysis,. Electrolysis Chem Libre.
From ptx-hub.org
Water electrolysis explained the basis for most PowertoX processes Electrolysis Chem Libre in galvanic cells, chemical energy is converted into electrical energy. The opposite is true for electrolytic cells. Compare the operation of electrolytic cells with that of galvanic cells. electrolysis is a process by which electrons are forced through a chemical cell, thus causing a chemical. in electrolysis, an electric current it sent through an electrolyte and into. Electrolysis Chem Libre.
From 2012books.lardbucket.org
Electrolysis Electrolysis Chem Libre in electrolysis, an electric current it sent through an electrolyte and into solution in order to stimulate the flow of. The opposite is true for electrolytic cells. Describe the process of electrolysis. Compare the operation of electrolytic cells with that of galvanic cells. By the end of this section you will be able to: Describe the process of electrolysis.. Electrolysis Chem Libre.
From www.scribd.com
Chem Lab Electrolysis PDF Electrolysis Chem Libre using electricity to force a nonspontaneous process to occur is electrolysis. Describe the process of electrolysis. in galvanic cells, chemical energy is converted into electrical energy. By the end of this section you will be able to: electrolysis is a process by which electrons are forced through a chemical cell, thus causing a chemical. The opposite is. Electrolysis Chem Libre.
From courses.lumenlearning.com
Electrolytes Chemistry for Majors Electrolysis Chem Libre electrolysis is a process by which electrons are forced through a chemical cell, thus causing a chemical. Compare the operation of electrolytic cells with that of galvanic cells. Describe the process of electrolysis. in electrolysis, an electric current it sent through an electrolyte and into solution in order to stimulate the flow of. using electricity to force. Electrolysis Chem Libre.
From socratic.org
Electrolysis Chemistry Socratic Electrolysis Chem Libre Describe the process of electrolysis. in galvanic cells, chemical energy is converted into electrical energy. Describe the process of electrolysis. electrolysis is a process by which electrons are forced through a chemical cell, thus causing a chemical. By the end of this section you will be able to: The opposite is true for electrolytic cells. Compare the operation. Electrolysis Chem Libre.
From courses.lumenlearning.com
Electrolysis General Chemistry Electrolysis Chem Libre Describe the process of electrolysis. using electricity to force a nonspontaneous process to occur is electrolysis. in galvanic cells, chemical energy is converted into electrical energy. electrolysis is a process by which electrons are forced through a chemical cell, thus causing a chemical. The opposite is true for electrolytic cells. in electrolysis, an electric current it. Electrolysis Chem Libre.
From mavink.com
Labelled Diagram Of Electrolysis Electrolysis Chem Libre Compare the operation of electrolytic cells with that of galvanic cells. in electrolysis, an electric current it sent through an electrolyte and into solution in order to stimulate the flow of. electrolysis is a process by which electrons are forced through a chemical cell, thus causing a chemical. By the end of this section you will be able. Electrolysis Chem Libre.
From chem.libretexts.org
5.6 Day 41 Electrolysis; Commercial Batteries Chemistry LibreTexts Electrolysis Chem Libre By the end of this section you will be able to: using electricity to force a nonspontaneous process to occur is electrolysis. in electrolysis, an electric current it sent through an electrolyte and into solution in order to stimulate the flow of. The opposite is true for electrolytic cells. Compare the operation of electrolytic cells with that of. Electrolysis Chem Libre.
From www.vecteezy.com
Electrolysis diagram experiment for education 1610466 Vector Art at Electrolysis Chem Libre Describe the process of electrolysis. The opposite is true for electrolytic cells. Compare the operation of electrolytic cells with that of galvanic cells. in electrolysis, an electric current it sent through an electrolyte and into solution in order to stimulate the flow of. using electricity to force a nonspontaneous process to occur is electrolysis. Describe the process of. Electrolysis Chem Libre.
From chem.libretexts.org
20.9 Electrolysis Chemistry LibreTexts Electrolysis Chem Libre Compare the operation of electrolytic cells with that of galvanic cells. using electricity to force a nonspontaneous process to occur is electrolysis. in electrolysis, an electric current it sent through an electrolyte and into solution in order to stimulate the flow of. The opposite is true for electrolytic cells. in galvanic cells, chemical energy is converted into. Electrolysis Chem Libre.
From www.youtube.com
Electrolysis Chem Video Textbooks Preview YouTube Electrolysis Chem Libre electrolysis is a process by which electrons are forced through a chemical cell, thus causing a chemical. Describe the process of electrolysis. By the end of this section you will be able to: in electrolysis, an electric current it sent through an electrolyte and into solution in order to stimulate the flow of. The opposite is true for. Electrolysis Chem Libre.
From byjus.com
What is Electrolysis in Chemistry? Electrolysis Chem Libre using electricity to force a nonspontaneous process to occur is electrolysis. in galvanic cells, chemical energy is converted into electrical energy. Compare the operation of electrolytic cells with that of galvanic cells. electrolysis is a process by which electrons are forced through a chemical cell, thus causing a chemical. Describe the process of electrolysis. By the end. Electrolysis Chem Libre.
From studyrocket.co.uk
Electrolysis GCSE Chemistry Science) OCR Revision Study Electrolysis Chem Libre Compare the operation of electrolytic cells with that of galvanic cells. By the end of this section you will be able to: in electrolysis, an electric current it sent through an electrolyte and into solution in order to stimulate the flow of. electrolysis is a process by which electrons are forced through a chemical cell, thus causing a. Electrolysis Chem Libre.
From melscience.com
“Electrolysis” experiment MEL Chemistry Electrolysis Chem Libre By the end of this section you will be able to: The opposite is true for electrolytic cells. in galvanic cells, chemical energy is converted into electrical energy. in electrolysis, an electric current it sent through an electrolyte and into solution in order to stimulate the flow of. electrolysis is a process by which electrons are forced. Electrolysis Chem Libre.
From thechemistrynotes.com
Quantitative Electrolysis Faraday's Law and Calculation Electrolysis Chem Libre in electrolysis, an electric current it sent through an electrolyte and into solution in order to stimulate the flow of. using electricity to force a nonspontaneous process to occur is electrolysis. Compare the operation of electrolytic cells with that of galvanic cells. electrolysis is a process by which electrons are forced through a chemical cell, thus causing. Electrolysis Chem Libre.
From studylib.net
Electrolysis Chemistry 40 Electrolysis Chem Libre electrolysis is a process by which electrons are forced through a chemical cell, thus causing a chemical. using electricity to force a nonspontaneous process to occur is electrolysis. Compare the operation of electrolytic cells with that of galvanic cells. Describe the process of electrolysis. Describe the process of electrolysis. in electrolysis, an electric current it sent through. Electrolysis Chem Libre.